Drug Safety Risk Calculator
This tool helps you assess the personalized safety risk of FDA-approved drugs from 2024-2025 based on your medical profile. It uses real-world data from the article to show potential risks and monitoring requirements.
What’s new in drugs? A look at 2024 and 2025’s biggest approvals
The FDA approved 50 new drugs in 2024 - the highest number since 2018. That’s not just a statistic. It’s a shift in how we treat serious, long-standing conditions. These aren’t minor tweaks to old medicines. These are entirely new ways of tackling disease - from Alzheimer’s to schizophrenia to anaphylaxis. And with each breakthrough comes a new set of safety questions.
Of those 50, nearly half (24) were first-in-class, meaning they work in a way no other approved drug does. That’s exciting for patients who’ve run out of options. But it also means doctors and patients are navigating uncharted territory. What happens when a drug targets a biological pathway never touched before? Side effects can be unpredictable. That’s why safety profiles are just as important as how well the drug works.
Alzheimer’s gets two new tools - but with serious risks
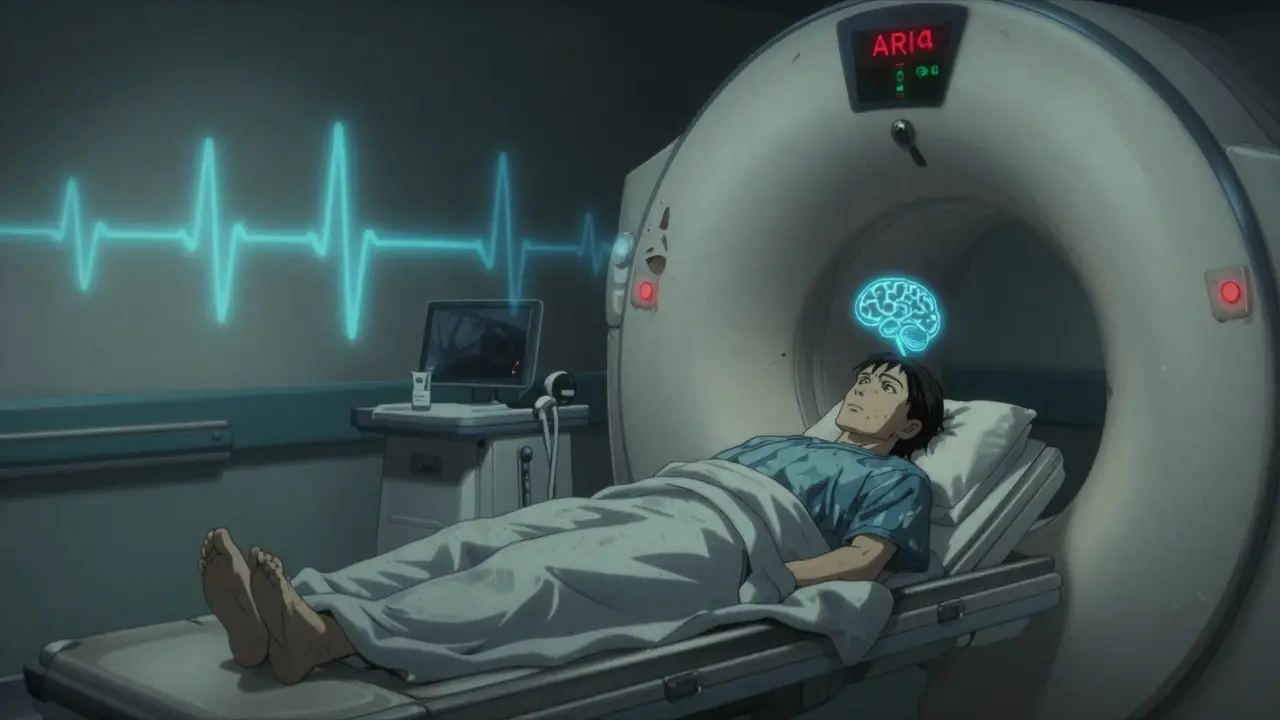
Donanemab-azbt (Kisunla) joined lecanemab (Leqembi) in 2024 as the second amyloid-targeting antibody for early Alzheimer’s. In clinical trials, it slowed cognitive decline by 35% over 18 months. That’s meaningful for someone worried about losing their memories. But it came with a catch: 24% of patients had ARIA - amyloid-related imaging abnormalities. That’s brain swelling or bleeding, detectable only by MRI. The placebo group? Just 2.9%.
Real-world data since approval is already showing something even more concerning: ARIA rates are 5-7 percentage points higher than in trials. That spike is strongest in people with two copies of the APOE ε4 gene - a known genetic risk factor. The FDA now requires strict monitoring through a Risk Evaluation and Mitigation Strategy (REMS) program. Patients must get regular MRIs. Doctors must be trained. This isn’t a drug you start on a hunch. It’s a treatment that demands careful, ongoing oversight.
Overdose reversal gets a new weapon
Nalmefene injection (Zurnai), approved in December 2024, is the first nasal spray opioid antagonist that lasts longer than naloxone. In trials, it kept working for 6.2 hours - more than double naloxone’s 2.1 hours. That’s huge. Opioid overdoses often come with multiple doses needed, especially with powerful fentanyl analogs. Zurnai’s longer action means fewer repeat doses, fewer trips to the ER, and less chance of a second overdose after the first wears off.
It also had fewer respiratory complications requiring extra help. That’s a win. But it’s not a magic bullet. It still requires training. And it doesn’t replace the need for emergency services. Still, for first responders and families, it’s a meaningful upgrade.
Anaphylaxis gets a needle-free option
Epinephrine nasal spray (Neffy) was approved in November 2024. For years, the only way to treat a life-threatening allergic reaction was a shot. Neffy changes that. In simulated use studies, 98% of untrained people could use it correctly - compared to 87% with auto-injectors. That’s a big deal. Panic, shaking hands, and fear can make injecting hard. A nasal spray? Much simpler.
But there’s a trade-off. It takes 15% longer to reach peak levels in the blood. For someone in full anaphylactic shock, every minute counts. Real-world reports show a 22% higher rate of treatment failure in severe cases. That means Neffy is great for mild-to-moderate reactions or as a first step - but not a replacement for an epinephrine shot in critical situations. Patients still need to carry both, at least for now.

Schizophrenia gets a new kind of treatment
For 27 years, schizophrenia treatment meant blocking dopamine. Then came Xanomeline and trospium chloride (Cobenfy) in September 2024. It’s the first drug in decades that targets muscarinic receptors instead. In trials, it improved psychosis scores by 34% - similar to older drugs - but with far fewer side effects. Nausea? 12%. Constipation? 8%. Compare that to the 25% and 18% rates with typical antipsychotics.
That’s huge. Weight gain, tremors, and sedation have kept many people from sticking with treatment. Cobenfy doesn’t cause those. But it does carry anticholinergic risks - dry mouth, blurred vision, constipation. The FDA requires patient education to help people manage these. It’s not a cure. But for many, it’s the first tolerable option in years.
Weight loss drugs are getting smarter
Tirzepatide (Zepbound) got a new use in December 2024: obstructive sleep apnea. The SURMOUNT-OSA trial showed a 46% drop in apnea events - thanks to a 4.9% weight loss. That’s not massive, but it’s enough to make a difference. For people with obesity-related sleep apnea, this could mean fewer CPAP machines and better sleep.
Wegovy (semaglutide) is heading in the same direction. In 2025, it’s expected to be approved for heart failure with preserved ejection fraction (HFpEF). The STEP-HFpEF trial showed improved quality of life and 13.3% weight loss. The side effects? Still mostly GI - nausea, vomiting, diarrhea. But no new risks. That’s important. GLP-1 drugs are now being tested for conditions far beyond weight loss: heart failure, kidney disease, even neurodegeneration. Their safety profile, while not perfect, is becoming better understood.
Other breakthroughs in 2024
- Palopegteriparatide (Yorvipath): For hypoparathyroidism. 89% of patients reached normal calcium levels without supplements. Side effects? Nausea (22%) and dizziness (15%) - much lower than older treatments.
- Sulopenem etzadroxil/probenecid (Orlynvah): A new oral antibiotic for bladder infections. 84% cure rate. No serious C. diff cases. A safe alternative to fluoroquinolones, which carry black box warnings for tendon rupture and nerve damage.
- Dupilumab (Dupixent): Approved for COPD. Reduced flare-ups by 29%. But 17% had injection site reactions. Not a game-changer for everyone, but helpful for a subset with high inflammation.
What’s coming in 2025?
The pipeline doesn’t stop. Over a dozen drugs have FDA review dates set for 2025. Here are the most notable:
- Cardamyst (etripamil): A nasal spray for sudden heart palpitations (PSVT). Converts rhythm in 74% of cases within 30 minutes. No serious heart risks. Just nasal discomfort.
- Elinzanetant: For menopause hot flashes. Reduces them by 52% - without hormone therapy’s stroke or blood clot risks. Side effects? Headache, dry mouth, constipation.
- Leqembi (subcutaneous): A needle-free version of the Alzheimer’s drug. Same effect, but injected under the skin. Easier for home use. Slightly more injection site redness - but no increase in brain swelling.
- Keytruda (subcutaneous): Cancer immunotherapy given as a shot instead of an IV. Reduces administration time by 74%. Infusion reactions drop from 5.2% to 0.8%. Big win for quality of life.
- Yutrepia (treprostinil): A new inhaled form for pulmonary hypertension. Reduces dosing frequency by 40%. But 22% report infusion pain - requires special training.
- Nipocalimab: For myasthenia gravis. First FcRn antagonist. Improves daily function. Infection risk? Only slightly higher than placebo.
Safety isn’t just about side effects - it’s about context
Dr. Rebecca Sarpong, one of the lead authors on the 2025 drug review, puts it simply: “These drugs are powerful, but they’re not one-size-fits-all.”
A drug that works great for a 55-year-old with no other conditions might be dangerous for someone with kidney disease or on multiple medications. That’s why the FDA now requires 24% of 2024 approvals to conduct long-term safety studies - up from 17% in 2023. These studies track real patients over years, not just months.
Early real-world data is already revealing gaps. For example, Neffy’s failure rate in severe anaphylaxis wasn’t predicted in trials. Kisunla’s ARIA rates are higher in real life. That’s why the FDA is working with the EMA and other agencies to share global safety data faster. No country can see everything alone.
And clinicians? A Sermo survey found 68% of primary care doctors asked for more training on at least one new drug from 2024. That’s not a failure - it’s a sign of responsible practice. We’re learning as we go.
What does this mean for you?
If you’re a patient: Don’t assume a new drug is automatically better. Ask: What’s the evidence? What are the real risks? Is this right for my body and lifestyle? Shared decision-making isn’t a buzzword - it’s essential.
If you’re a caregiver: Understand the monitoring requirements. Kisunla needs MRIs. Cobenfy needs education on anticholinergic effects. Neffy needs a backup plan for severe reactions. Knowledge is your tool.
If you’re a clinician: Stay curious. Use the FDA’s REMS database. Attend pharmaceutical updates. The pace of innovation is fast. Your job isn’t just to prescribe - it’s to understand.
These drugs represent hope. But hope without caution can be dangerous. The balance between innovation and safety has never been more critical. And we’re all learning how to walk that line - together.


Tobias Mösl
March 3, 2026 AT 06:04Ethan Zeeb
March 4, 2026 AT 22:05Darren Torpey
March 6, 2026 AT 19:45Lebogang kekana
March 7, 2026 AT 15:07Raman Kapri
March 9, 2026 AT 11:56Megan Nayak
March 10, 2026 AT 11:19Tildi Fletes
March 11, 2026 AT 13:27Pankaj Gupta
March 12, 2026 AT 04:04Zacharia Reda
March 13, 2026 AT 23:25Stephen Vassilev
March 14, 2026 AT 04:14Mike Dubes
March 14, 2026 AT 10:50John Cyrus
March 15, 2026 AT 02:18Jessica Chaloux
March 17, 2026 AT 00:11Mariah Carle
March 17, 2026 AT 10:29Renee Jackson
March 18, 2026 AT 10:44